The drug products are tested under various environmental conditions to collect data regarding the stability of drug products.
Pharmaceutical drug products are stored under various ranges of temperature & humidity & are tested to check the safety & efficacy of that drug product after a specified period of time.
The temperature & humidity of all the countries is not the same. That is why the ICH has divided the whole world into various climatic zones.
Importance Of Climatic
Stability zones play an important role in collecting stability data of a drug product.
- One must have to collect stability data of new drug products according to the climatic zone of the country where it is going to be registered.
- For export of a drug product in a specific country we must collect stability data according to the zone of that country.
- Storage conditions for various pharmaceutical drug products are determined by testing the product in various climatic conditions.
Climatic Zones For Stability Studies
The world is divided into four climatic zones & zone IV is further subdivided into two zones which are IVa & IVb so by adding by division we have 5 ICH climatic zones.
The numbering of climatic zones for stability studies is given below,
- Zone I
- Zone II
- Zone III
- Zone IVa
- Zone IVb
The names of above mentioned climatic zones are given below
| Zone | Zone Name/Climate |
| Zone I | Temperate |
| Zone II | Mediterranean/
Subtropical |
| Zone III | Hot Dry |
| Zone IVa | Hot Humid/Tropical |
| Zone IVb | Hot Higher Humidity |
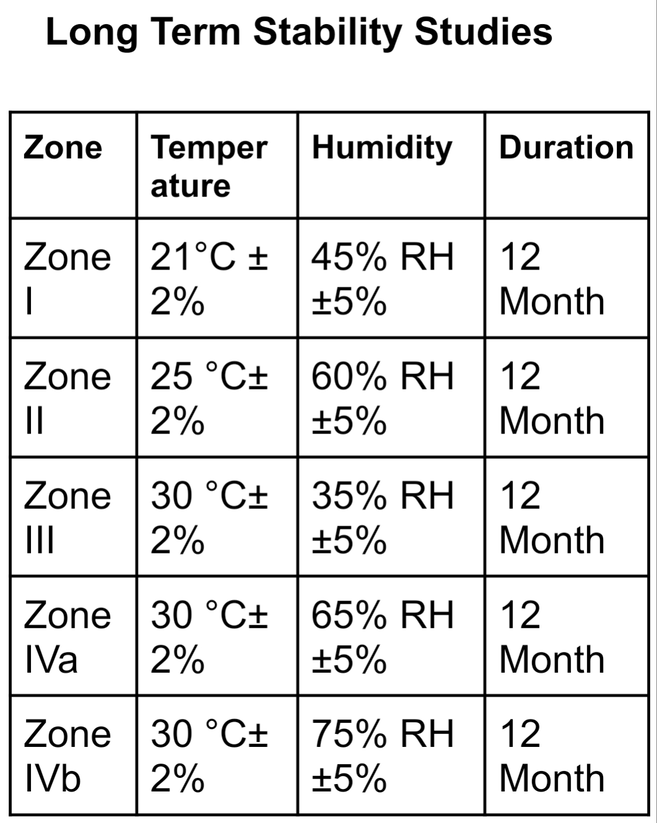
Following are the long-term stability study conditions for above mentioned ICH stability zones
Long-Term Stability Studies
| Zone | Temperature | Humidity | Minimum time at submission |
| Zone I | 21°C ± 2% | 45% RH ±5% | 12 Month |
| Zone II | 25 °C± 2% | 60% RH ±5% | 12 Month |
| Zone III | 30 °C± 2% | 35% RH ±5% | 12 Month |
| Zone IVa | 30 °C± 2% | 65% RH ±5% | 12 Month |
| Zone IVb | 30 °C± 2% | 75% RH ±5% | 12 Month |
Countries According To Climatic Zones
Following is the division of main countries according to their climatic zones
| Name | Zone |
| UK |
Zone I |
| North Europe | |
| Germany | |
| Russia | |
| Canada |
| Name | Zone |
| USA |
Zone II |
| China | |
| Iran | |
| Japan | |
| Australia |
| Name | Zone |
| Iraq |
Zone III |
| Jordan | |
| Botswana |
| Name | Zone |
| Pakistan |
Zone IV |
| India | |
| Bangladesh | |
| Omam | |
| EUE | |
| Indonesia |
Which country in IVa & IVb?
India in zone 4b
For hong kong what is the zonal stability to consider whether it is Zone 4 or Zone 4B?